Sion, Mumbai, Maharashtra
- GST NO. : 27AQZPP9186B1ZZ
View Mobile Number
| Business Type | Manufacturer, Exporter, Supplier, Retailer |
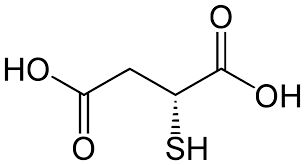
| Appearance (Colour) | White to off-white |
| Appearance (Form) | Crystalline compound |
| Assay | min. 99% |
| Click to view more | |
Product Details
Melting Point
150 - 152°C
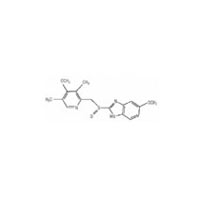
Thiomalic Acid (Mercaptosuccinic Acid) pure, 99%
CAS NO 70-49-5
Molecular Formula : C4H6O4S
Molecular Weight : 150.16
Part B
Storage : Room Temperature
HSN Code : 29171990
Looking for "Thiomalic Acid" ?
Litre
Explore More Products